Electrocatalysis
We are working on investigation of the activity, stability and selectivity of the electrocatalyst materials towards electrocatalysis reactions such as oxygen reduction reaction (ORR), CO2 reduction reaction, and H2 productions. We work on synthesis and characterization of novel-metal-free electrocatalysts using advanced in-situ/operando technique to provide a solid understanding and sound foundation for development of sustainable electrocatalysts for highly effective energy conversion systems.
Fuel Cells/Oxygen Reduction Reaction
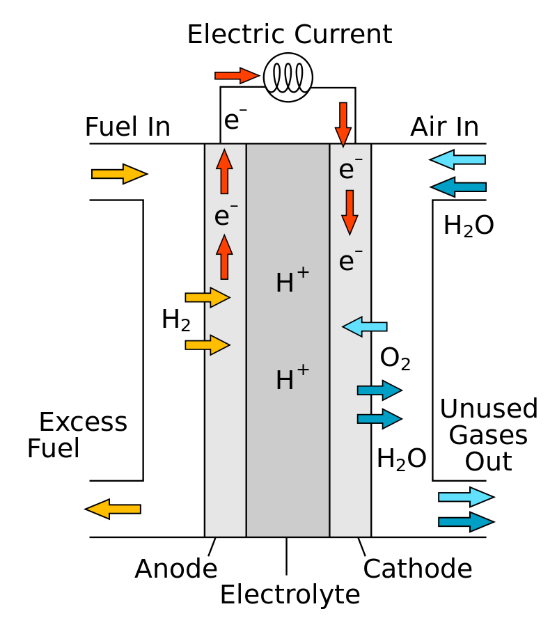
Figure 1 Scheme of a proton-conducting fuel cell. Image referred from: https://en.wikipedia.org/wiki/Fuel_cell
Fuel cells are promising candidates for clean and efficient electricity generation. The ORR reaction at the cathode is one of the main rate-determine steps due to its sluggish efficiency. At the current state of technology, Pt and Pt-alloys are the most practical catalysts used to speed the ORR reaction at fuel cell cathode. However, their high cost and poor stability hinders the further development of the fuel cell devices.
H2 evolution
Hydrogen for fuel cells to be used in transportation or industrial sector is an important approach to avoid future CO2 emissions. Today, only 4% of hydrogen is produced from electrolysis while other lower-cost but greenhouse gases emissive methods are preferred, such as steam reforming of coal or natural gas.
CO2 reduction reaction
CO2 reduction via electrochemical routes (CO2RR) attracted a lot of attention recently, being considered an alternative to renewable energy crisis. Along the years, different electrocatalysts have been used for CO2RR, very common being noble metals (Ag, Au, Pd). The main challenge for this process consists in finding inexpensive, selective and stable catalysts, operating at low overpotentials.